Executive Summary
Front-of-pack nutrition labeling systems were designed to solve a real problem: most consumers cannot interpret raw nutrient data at the point of purchase. In several jurisdictions, simplified visual signals have measurably influenced purchasing patterns and product reformulation. Yet the architecture of compression — collapsing multiple nutritional dimensions into a single score, rating, or warning — requires normative weighting decisions that differ across regulatory contexts. The same product can receive materially different evaluations under different national systems, not because any system is poorly designed, but because each embeds distinct dietary priorities.
At least 44 countries have now introduced front-of-pack labeling policies, with no interoperability layer between them. This paper examines the structural consequences of that fragmentation — for public health governance, for cross-border trade, and for the consumers these systems were built to serve.
1. The Promise
The premise behind front-of-pack nutrition labeling is sound. Nutrient declaration tables — lists of energy, fat, sugar, sodium, protein, and other values per serving or per 100 grams — provide comprehensive information. But decades of consumer research have confirmed that most shoppers do not read them, and among those who do, many struggle to interpret what the numbers mean in context. The information exists. The interpretation does not.
Front-of-pack systems were designed to close that gap. By placing a simplified visual signal on the front of a product — a letter grade, a star rating, a colour code, or a warning symbol — regulators aimed to make nutritional quality legible at the moment of decision. The ambition was population-level: not to educate every individual consumer in nutritional science, but to shift aggregate purchasing patterns toward healthier options.
In several jurisdictions, the evidence suggests this ambition has been partially realised. Chile's 2016 food labeling and marketing law — which combined octagonal warning labels with restrictions on child-directed marketing and school food sales — produced the strongest population-level evidence to date. An interrupted time-series analysis covering three years of post-implementation data found sustained reductions in purchases of products carrying warning labels: 37% for sugar, 22% for sodium, 16% for saturated fat, and 23% for calories. Sugar reduction was greatest in beverages, at 54%. Notably, these effects were consistent across socioeconomic groups.
In Europe, the adoption of Nutri-Score — a five-colour, five-letter scale (A through E) — has been associated with measurable product reformulation in France and Belgium, as manufacturers adjusted recipes to achieve more favourable ratings. In Australia and New Zealand, the voluntary Health Star Rating system prompted similar reformulation activity, particularly in the sugar and sodium content of processed foods, as documented in the system's five-year review.
The question this paper raises is not whether that promise has value. It does. The question is what happens architecturally when the method of simplification — compression of multidimensional nutritional data into a single scalar output — is replicated independently across dozens of jurisdictions, each embedding different assumptions, for products that circulate in a global trade environment.
2. The Structural Problem
Every front-of-pack scoring system performs the same fundamental operation: it takes a set of independent nutritional variables and collapses them into a single output — a letter, a number of stars, a set of warning octagons, or a grade. Regardless of format, the operation is the same: dimensional reduction. Dimensional reduction is not inherently problematic, but it carries an inherent constraint: the reduction requires weighting. Which nutritional dimension matters more — saturated fat or added sugar? Should fibre content offset sodium levels? Does the presence of protein compensate for high energy density? Every scoring algorithm answers these questions, and every algorithm answers them differently.
These are not technical calibration choices. They are normative decisions that reflect specific dietary priorities, epidemiological profiles, and policy objectives. France's Nutri-Score penalises saturated fat and energy density, reflecting concerns about cardiovascular disease. Chile's warning labels set absolute thresholds for sugar, sodium, saturated fat, and calories, reflecting an aggressive stance on obesity and metabolic disease. Australia's Health Star Rating balances negative nutrients against positive ones (protein, fibre, fruit/vegetable content). Singapore's Nutri-Grade applies exclusively to beverages, reflecting a targeted policy decision to address sugar-sweetened drink consumption.
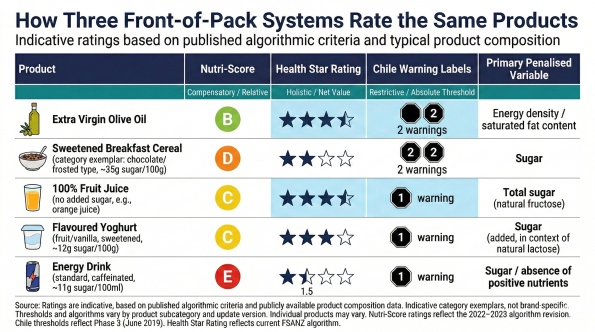
Each system is internally coherent. Each reflects legitimate public health reasoning. But when the same product passes through multiple systems, the divergence becomes visible. Under the original Nutri-Score algorithm, extra virgin olive oil received a C rating — penalised by its high fat content despite its well-documented role in healthy dietary patterns. Under Australia's Health Star Rating, the same product scores approximately 4 out of 5 stars. The Nutri-Score Scientific Committee acknowledged this tension and revised the algorithm through an update cycle spanning 2022–2023, moving olive oil from C to B. But even after revision, a single letter cannot express the full nutritional and cultural dimensionality of a product whose health value depends on how it is used, how much is consumed, and what it replaces in a given dietary pattern. The revision itself demonstrates the structural point: single-score systems require ongoing algorithmic adjustment as edge cases surface, because the compression inevitably produces outcomes that conflict with broader nutritional evidence.
“Interpretation has not disappeared; it has shifted — from the consumer at the shelf to the algorithm that determines the score.”
This constraint generates several downstream effects. First, it creates conditions for halo effects: a favourable score signals the absence of penalised attributes, but does not signal the presence of positive ones. Because most single-score systems weight nutrient composition rather than degree of processing, products with favourable nutrient profiles may still be classified as ultra-processed under independent frameworks such as NOVA — illustrating how compression can obscure dimensions that lie outside the scoring model's chosen variables.
Second, it creates incentives for selective reformulation. Manufacturers respond to scoring systems by reformulating products to improve their ratings, but this reformulation is uneven — concentrated on the nutrients most heavily weighted by the algorithm. Sodium and sugar content show the greatest reformulation response; saturated fat shows less.
The governance implication is significant. When a numerical score performs the act of interpretation on behalf of a consumer, the interpretive authority — and the accountability for the weighting decisions embedded in that interpretation — shifts from the individual to the algorithm designer. The public health authority does not merely disclose information; it embeds a hierarchy of nutritional values into a mandatory or quasi-mandatory signal. The shift is structural, and largely invisible to the consumer who encounters only the simplified output.
2A. Why This Is Not a Design Failure
Front-of-pack labeling systems were designed to achieve domestic public health objectives, and on those terms, several have produced measurable results. Nutrition policy reflects legitimate sovereign priorities. Dietary risk profiles differ across populations. A system designed to address Chile's sugar consumption patterns will differ structurally from one designed for Mediterranean dietary contexts, and both will differ from one designed for a high-processed-food market like Australia's.
Cross-border harmonisation of these systems was neither politically intended nor necessarily desirable. The European Commission's experience illustrates this: its Farm-to-Fork commitment to a harmonised front-of-pack system (2020) has not progressed to a formal legislative proposal as of early 2026, following a public consultation that revealed deep political divisions. Several member states — led by Italy — formally opposed Nutri-Score at the Council level and introduced their own NutrInform Battery system. Italy introduced a constitutional amendment proposal in May 2024 to protect traditional food products from algorithmic scoring.
The strongest defence of the current landscape is straightforward: cross-border interoperability was never the design objective. The objective was to reduce dietary harm within a single national population. The structural consequence emerges when dozens of domestically coherent systems operate simultaneously within a globally integrated trade environment — without a coordination mechanism between them. The limitation is architectural, not intentional.
3. Evidence and Examples
The following regional survey illustrates the scope of system diversity and the structural incoherence that results when multiple independent systems operate simultaneously in global trade contexts.
Europe
Nutri-Score was developed by French public health researchers and adopted by France in 2017, followed by Belgium, Germany, Luxembourg, the Netherlands, Spain, and Switzerland. Its algorithm was revised through an update cycle spanning 2022–2023, modifying the treatment of oils, fats, fish, dairy, and whole grains. Despite adoption by seven countries, the system remains voluntary in all of them, and the European Commission's effort to mandate a harmonised EU-wide system has stalled indefinitely.
The political dynamics are instructive: Italy, Greece, the Czech Republic, Hungary, Romania, Latvia, and Cyprus have formally opposed Nutri-Score at the EU Council level, arguing that it penalises traditional food products — particularly olive oil, aged cheeses, and cured meats — that carry geographical indications and form the basis of dietary patterns recognised by UNESCO as intangible cultural heritage. The debate has become a proxy for broader tensions between public health standardisation and trade protection of agricultural heritage.
Oceania
Australia and New Zealand adopted the Health Star Rating in 2014 as a voluntary, government-endorsed interpretive system. Its five-year review, conducted by Food Standards Australia New Zealand (FSANZ) in 2019, found evidence of reformulation activity but also identified structural concerns: the original algorithm under-penalised sugar and produced counterintuitive results for certain product categories, including energy drinks that received moderate star ratings despite high sugar and caffeine content. The review recommended ten specific algorithmic adjustments.
Participation remains voluntary, and adoption rates vary significantly across product categories, with larger manufacturers more likely to display the rating than smaller producers — creating an uneven information landscape on retail shelves.
The Americas
Chile's Ley 20.606 (2016) represents the most comprehensive and most studied front-of-pack intervention globally. Its strongly-evidenced effects — 37% reduction in sugar purchases, 22% sodium, 16% saturated fat, 23% calories — reflect the combined policy ecosystem of warning labels, marketing restrictions, and school food sales bans. This distinction matters: simplified labeling achieves its strongest effects when embedded in a broader regulatory architecture.
Mexico (2020), Uruguay (2018), Colombia (2022), and Brazil (2022) have adopted similar warning-label approaches. In the United States, the FDA proposed a new front-of-pack labeling scheme in January 2025, focusing on saturated fat, sodium, and added sugars — presenting nutrient-specific levels (Low, Medium, High) rather than a single summary grade, illustrating that jurisdictions are diverging not only on thresholds but on the degree of compression itself.
Asia
Singapore's Nutri-Grade system, mandatory for pre-packaged beverages since December 2022, represents an instructive design choice: it restricts its scope to a single product category rather than attempting a universal score. Within that narrow scope, it has produced measurable results — 71% of pre-packaged beverages now fall in the A or B category, up from 37% in 2017, and household purchases of C and D-rated beverages declined by approximately 90 mL per household per day. The narrower the product scope, the more coherent the compression.
Japan's Foods with Function Claims (FFC) system, launched in 2015 and amended in 2024, represents a structurally different approach: it allows manufacturers to make specific functional claims based on scientific evidence, without pre-market approval, leaving interpretation with the consumer rather than embedding it in an algorithm. India's FSSAI proposed a star-based Indian Nutrition Rating that attracted over 14,000 public comments and has since indicated its intention to withdraw the 2022 draft approach and reopen consultation — illustrating that the compression debate remains unresolved even within individual jurisdictions.
GCC (Contextual Note)
The Gulf Cooperation Council states, as major food importers, receive products bearing front-of-pack labels designed for other regulatory contexts. A product carrying a Nutri-Score label may sit on a shelf in Abu Dhabi alongside one carrying an Australian Health Star Rating and another with no front-of-pack information at all. No GCC-specific interpretive front-of-pack system currently exists. For importing economies, the fragmentation of source-country labeling systems creates an ambient information environment that is neither coherent nor absent — it is simply uncoordinated. Interpretive inconsistency arrives embedded in packaging, without any sovereign calibration of the weighting logic by the importing jurisdiction.
4. Implications
The structural fragmentation documented in the preceding sections has distinct consequences for different actors in the food system.
For nutrition and public health professionals, the tension is between domestic effectiveness and global incoherence. Within a single jurisdiction, a well-designed front-of-pack system can shift purchasing patterns and incentivise reformulation. Across jurisdictions, the same product may signal health in one market and trigger a warning in another — not because the product changed, but because the interpretive framework changed. This creates a governance environment in which the meaning of “healthy” is jurisdictionally contingent, shaped as much by algorithmic design as by nutritional evidence.
For trade agencies and export promotion bodies, the consequence is operational. A producer serving five or more markets currently maintains parallel documentation systems with no structured interface between them. A cereal manufacturer exporting to France, Mexico, and Australia may maintain three separate front-of-pack compliance models — because no translation layer exists that preserves the underlying nutritional record while rendering it through multiple national interpretive lenses. Unlike many forms of regulatory divergence, front-of-pack interpretive systems operate as algorithmic evaluation layers rather than static disclosure rules. Divergence therefore affects not only label compliance but formulation incentives and perceived product meaning in-market.
For policymakers and regulators, the governance question is structural. When a scoring algorithm performs interpretation on behalf of a consumer, the accountability for the weighting decisions embedded in that algorithm rests with its designers. Yet consumers encountering a score on a retail shelf have no visibility into what was weighted, what was excluded, or how the same product would be evaluated under a different system. The accountability gap is not a function of any individual system's transparency — several publish their algorithms openly — but of the structural shift from consumer interpretation to algorithmic interpretation at population scale.
For institutional procurement, the operational challenge is comparison. A hospital system, school district, or government procurement body sourcing food products from multiple countries cannot meaningfully compare items across scoring regimes. No structured mechanism currently exists to translate between systems or to enable cross-regime product comparison for institutional buyers.
5. Toward Structural Alternatives
The evidence reviewed in Section 1 confirms that simplified nutrition communication has measurable public health value. What the preceding analysis suggests is that the architecture of compression carries structural constraints that become increasingly consequential as these systems proliferate across a globally interconnected food trade environment.
Any structural alternative would need to satisfy several architectural requirements. It would need to retain the original dimensional data alongside whatever simplified rendering is applied, rather than discarding the source information in favour of the score. It would need to allow multiple interpretive lenses to coexist for the same product. It would need to enable cross-border translation between systems without requiring global harmonisation — permitting sovereign variation in interpretation while providing an interpretive interface that makes divergence legible rather than invisible. And it would need to separate the act of disclosure — making product information available — from the act of interpretation — assigning meaning to that information.
This separation is already standard practice in other governance domains: financial reporting requires structured data disclosure while multiple analytical models coexist; pharmaceutical documentation mandates comprehensive data while regulatory interpretation varies by jurisdiction; environmental disclosure frameworks publish emissions data that multiple rating systems then evaluate independently. Nutrition information governance differs in that the act of disclosure and the act of interpretation are compressed into a single output.
Several developments suggest movement in this direction. The Codex Alimentarius Commission adopted two new guidelines in November 2024: CXG 105-2024, on the use of technology to provide food information, and CXG 104-2024, on the provision of food information for products sold via e-commerce. Both establish principles for technology-enabled food information that go beyond what physical labels can carry. The GS1 Digital Link standard, with its “Sunrise 2027” deadline for industry adoption of 2D barcode systems, creates technical infrastructure through which structured product data could be accessed dynamically rather than compressed into a static on-pack symbol. The European Union's Digital Product Passport discussions point toward regulatory models that require structured data availability rather than simplified labeling as the primary information mechanism.
Digital disclosure does not automatically resolve equity constraints. Smartphone access, data costs, language availability, and digital literacy vary materially across populations. Any digitally enabled approach would need a default shelf-level rendering that works for low-literacy and low-connectivity settings, with richer detail available via optional access pathways. The architectural objective is not to replace simple labels, but to prevent simplified renderings from becoming the only legible representation of the nutritional record.
This paper does not propose a specific model. It outlines structural requirements that any viable alternative would need to satisfy.
References
- World Health Organization. Guiding Principles and Framework Manual for Front-of-Pack Labelling for Promoting Healthy Diets. Geneva: WHO, 2019.
- Taillie LS, Bercholz M, Popkin B, Rebolledo N, Reyes M, Corvalán C. "Decreases in Purchases of Energy, Sodium, Sugar, and Saturated Fat 3 Years after Implementation of the Chilean Food Labeling and Marketing Law: An Interrupted Time Series Analysis." PLOS Medicine 21(9): e1004463. September 2024.
- Taillie LS, et al. "An Evaluation of Chile's Law of Food Labeling and Advertising on Sugar-Sweetened Beverage Purchases from 2015 to 2017." PLOS Medicine 16(12): e1003015. February 2020.
- Nutri-Score: impact on reformulation activity documented in European Commission, Impact Assessment on Harmonised Front-of-Pack Nutrition Labelling — Public Consultation Summary Report. Brussels: European Commission, 2022.
- Food Standards Australia New Zealand (FSANZ). Health Star Rating System Five Year Review Report. Canberra: FSANZ, May 2019.
- European Scientific Committee of Nutri-Score. Reports on Changes to the Algorithm for Solid Foods and Beverages. Update cycle 2022–2023. See also: Sarda B, et al. "Consistency of the Initial and Updated Version of the Nutri-Score Nutrient Profile Model." Journal of Nutrition 154(4): 1229–1240. 2024.
- Forde H, et al. "Front-of-Package Nutrition Labeling and Its Impact on Food Industry Practices: A Systematic Review." Nutrients 15(11): 2630. 2023.
- European Commission. Farm-to-Fork Strategy: commitment to harmonised FOP nutrition labelling (2020). Public consultation December 2021 – March 2022 (3,225 responses). No formal legislative proposal tabled as of March 2026.
- Italian government opposition to Nutri-Score: NutrInform Battery alternative (2020); seven-country coalition statement (2020); constitutional amendment proposal (May 2024). Sources: Euronews Health; Italian Food News; European Parliament Legislative Train Schedule.
- Voo JYH, et al. "Impact of Singapore's Nutri-Grade Labels on Purchases." International Journal of Behavioral Nutrition and Physical Activity 20(1): 27. 2023. Also: Singapore Health Promotion Board implementation data, 2022–2024.
- Japanese Consumer Affairs Agency. Foods with Function Claims (FFC) System. Launched April 2015; amended September 2024. Market data: NutraIngredients, July 2024.
- Food Safety and Standards Authority of India (FSSAI). Draft notification on Indian Nutrition Rating (front-of-pack, 0.5–5.0 stars). Originally published 2022; FSSAI indicated intention to withdraw in Supreme Court filings, citing need for further research and stakeholder consensus.
- Codex Alimentarius Commission. Guidelines on the Use of Technology to Provide Food Information (CXG 105-2024). Adopted at the 47th Session, November 2024.
- Codex Alimentarius Commission. Guidelines on the Provision of Food Information for Pre-Packaged Foods to be Offered via E-Commerce (CXG 104-2024). Adopted November 2024.
- GS1. GS1 Digital Link Standard. 2024. Industry adoption deadline ("Sunrise 2027") for 2D barcode compliance.
- Afroza U, Abrar AK, Nowar A, Sobhan SMM, Ide N, Choudhury SR. "Global Overview of Government-Endorsed Front-of-Pack Nutrition Labelling Policies of Packaged Foods: A Document Review." Frontiers in Public Health 12: 1426639. November 2024.
- U.S. Food and Drug Administration. "Food Labeling: Front-of-Package Nutrition Information." Proposed Rule, 90 FR 5426. Federal Register, January 16, 2025. Docket: FDA-2024-N-2910.